By Tina Lidén Mascher
Most sponsors feel the pressure: hit milestones, hold budget, keep recruitment moving. At the same time, patient safety and data integrity can’t budge. The way through is not more documents; it’s clear oversight and risk‑based focus.
Start with what GCP actually asks of the sponsor
You can outsource tasks, not accountability. ICH E6(R3) Section 3.9.5 is unambiguous: The range and extent of oversight measures should be fit for purpose and tailored to the complexity of and risks associated with the trial. The selection and oversight of investigators and service providers are fundamental features of the oversight process. Oversight by the sponsor includes quality assurance and quality control processes relating to the trial-related activities of investigators and service providers.
ICH E6(R3) Section 3.10 adds: The sponsor should implement an appropriate system to manage quality throughout all stages of the trial process.
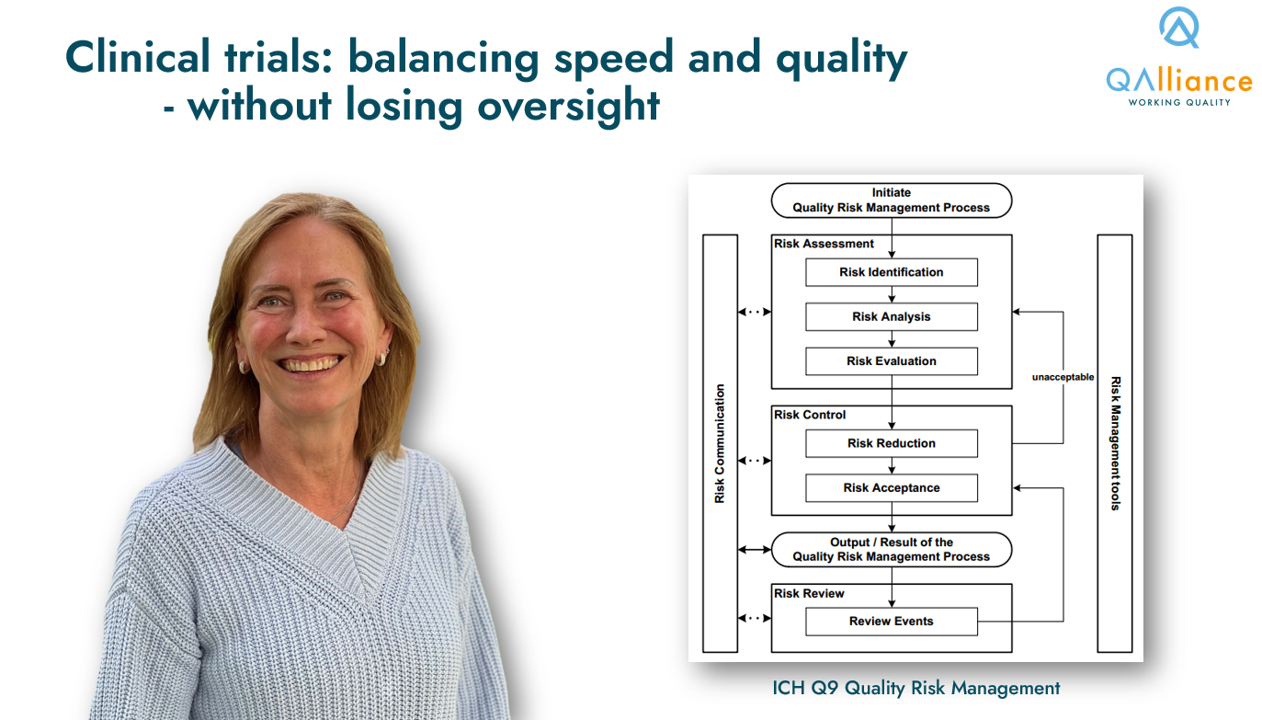
Focus on the risks, not the noise (Risk-based Quality Management)
When implementing such oversight system, we identify critical quality factors, critical data and critical processes. We agree on simple checks throughout the trial, and document the logic. This aligns with ICH Q9: be explicit about assumptions you use, and use relevant data to make decisions.
Our practical checklist for sponsor oversight
- Oversight plan: who looks at what, how often, where it’s recorded.
- A few CRO KPIs (Key Performance Indicators) that actually can make a difference (e.g., protocol deviations on critical processes, data timeliness on primary endpoints).
- Short monthly reviews, and if needed adjust oversight focus based on evidence collected.
- Keep the file inspection‑ready as a by‑product of work, not a separate project.
Fewer surprises, faster decisions, and documentation that actually reflects reality.
If you’re wrestling with the oversight of your ongoing or upcoming study, send us a message and we’ll help your team to get unstuck.